The correct staging of diabetic retinopathy (DR) is crucial to quantifying the level of pathology, which enables accurate risk assessment for disease progression and when to refer a patient to a retina specialist for treatment consideration. The Diabetic Retinopathy Severity Scale (DRSS), which comes from the Early Treatment Diabetic Retinopathy Study, has been readapted into the International Clinical Diabetic Retinopathy (ICDR) staging system, which is universally recognized and used widely in clinical practice. The ICDR is broken down into 2 main categories: nonproliferative diabetic retinopathy (NPDR) and proliferative diabetic retinopathy (PDR).1,2
Nonproliferative Diabetic Retinopathy Staging
Mild NPDR and Recommendation for Management
This is characterized by the presence of at least 1 microaneurysm or retinal hemorrhage (Figure 1). Microaneurysms represent outpouching of the retinal capillary walls due to weakening from pericyte loss. Hemorrhages result when the capillary wall ruptures within the inner nuclear or outer plexiform layer of the retina, therefore forming a characteristic dot/blot appearance.3 Fluorescein angiography (FA) is not indicated at this stage and color photos, while not necessary, are often useful for patient education regarding the importance of complying with their follow-up appointment and monitoring disease progression.
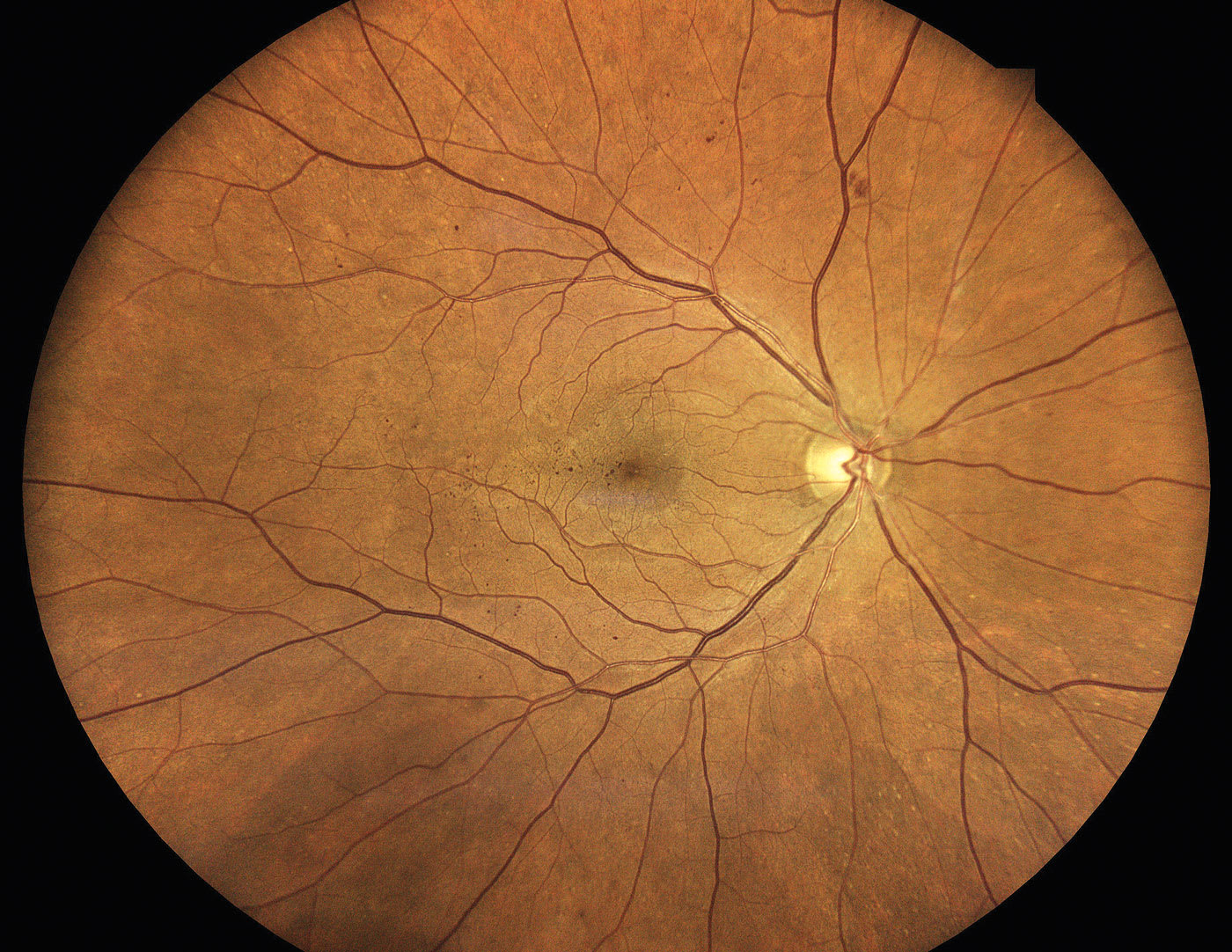
These patients should receive a repeat dilated exam in 1 year because approximately 5% to 10% of these patients’ NPDR will worsen in that time.4,5 Additionally, as with any stage of retinopathy, communication with the patient’s primary care doctor, notifying him or her of the presence of retinopathy and the importance of systemic disease management, as that can affect progression, is advised.
Moderate NPDR and Recommendation for Management
This is characterized by an increasing number of microaneurysms and hemorrhages, as well as the emergence of cotton wool spots, venous beading, and intraretinal microvasculature abnormalities (IRMAs) to a mild degree, less than that of severe NPDR (Figure 2). Cotton wool spots are fluffy, white retinal lesions that represent focal ischemia. Venous beading is a localized irregular sausage-like dilation of the vein.
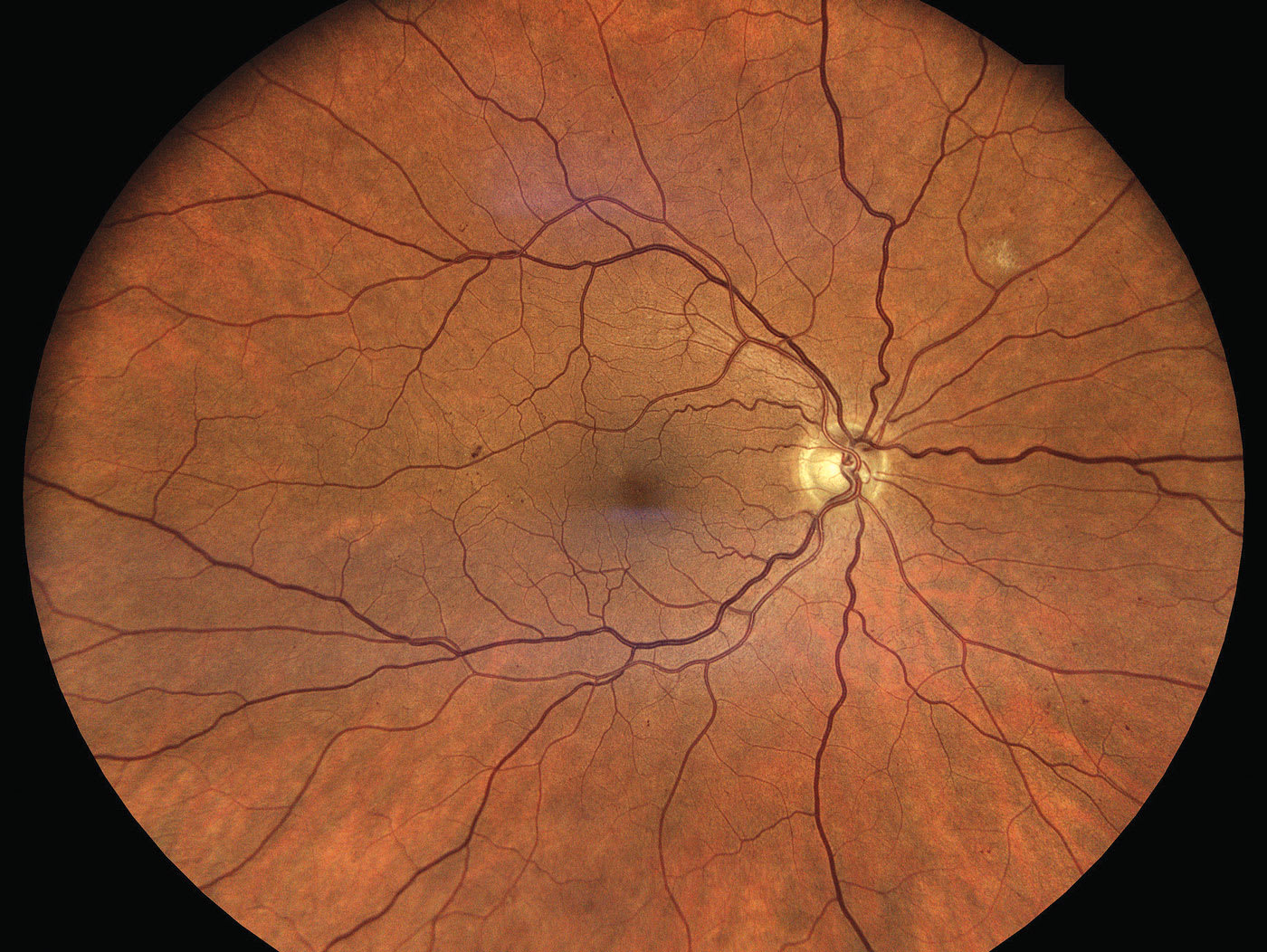
Intraretinal microvasculature abnormalities represent shunt vessels that have formed due to retinal nonperfusion. They can be distinguished from true neovascularization by FA in that IRMAs don’t leak dye, but true neovascularization does.
These patients should receive a repeat dilated exam within 6 months. As many as 16% of patients who have moderate NPDR progress to PDR within 4 years.4 Fluorescein angiography is not needed. Color fundus photos are a tool for monitoring progression and can be used for patient education to enhance compliance to the follow-up appointment.
Severe or Very Severe NPDR and Recommendation for Management
Severe NPDR is characterized by the “4-2-1” rule; that is, when all 4 quadrants of the retina have marked microaneurysms or hemorrhages, 2 quadrants have venous beading, or 1 quadrant demonstrates prominent IRMA (Figure 3). If 2 of the above criteria are met, the patient is classified as having very severe NPDR.6
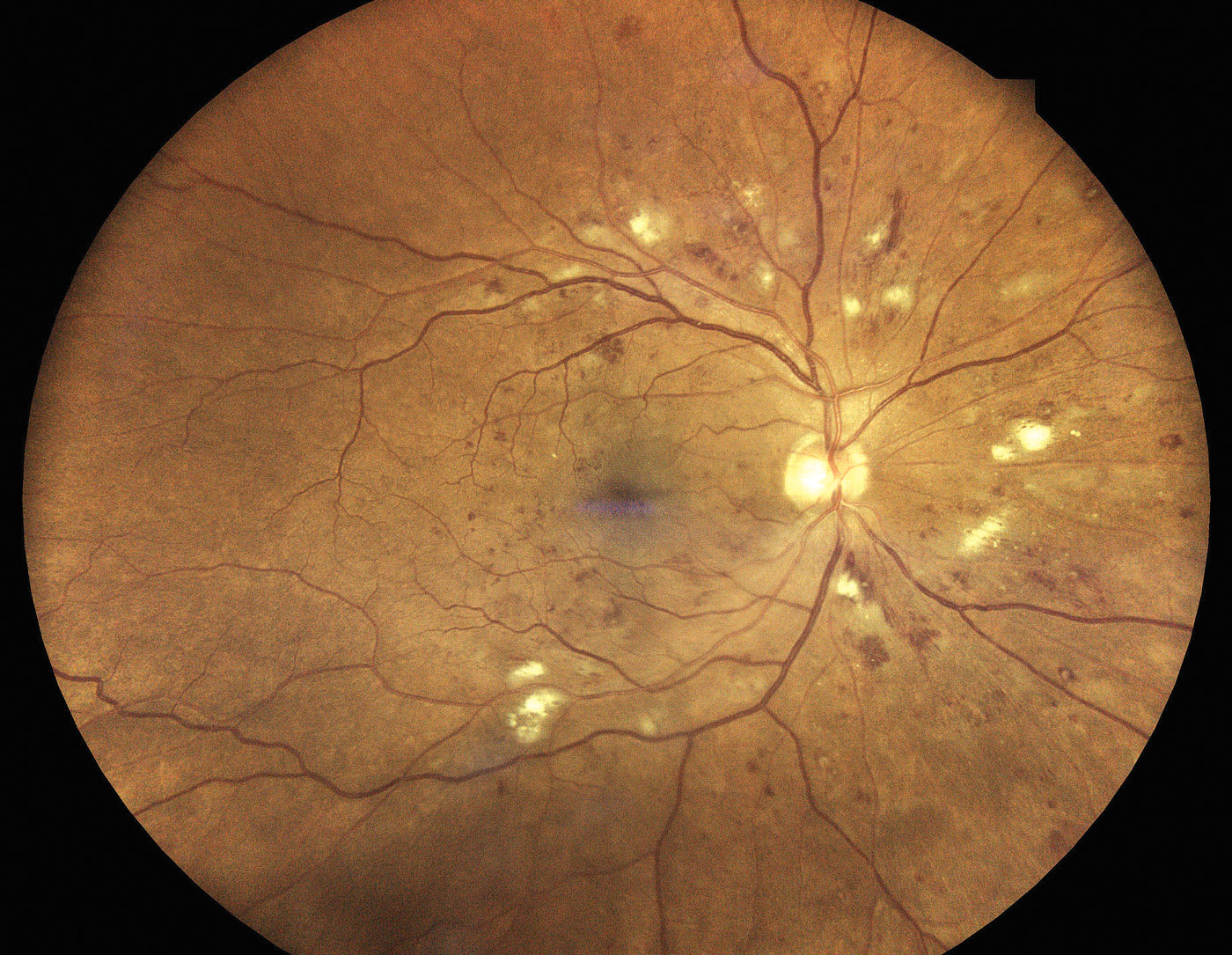
Approximately half of patients who have severe NPDR and nearly 75% of patients who have very severe NPDR will progress to PDR within 1 year.7 Therefore, these patients need to be followed every 2 to 3 months. Further, due to this high risk of disease progression, it makes sense to consider referral to a retina specialist. Notably, the disease is often found to be more advanced with widefield FA, revealing marked peripheral ischemia or occult neovascularization, and prompting the retina specialist to intervene with serial anti-VEGF.
PDR Staging and Management
This is characterized by neovascularization, vitreous hemorrhage, and/or preretinal hemorrhage. Neovascularization can present on or within 1 disc diameter of the optic nerve, called neovascularization of the disc (NVD), or elsewhere in the retina, known as neovascularization elsewhere (NVE; Figures 4 and 5).6 High-risk PDR occurs when the area of NVD is greater than one-fourth to one-third disc diameter in size, or any NVD that has an associated vitreous or preretinal hemorrhage, or NVE that is greater than one-half a disc diameter that has an associated vitreous or preretinal hemorrhage (Figure 6).7
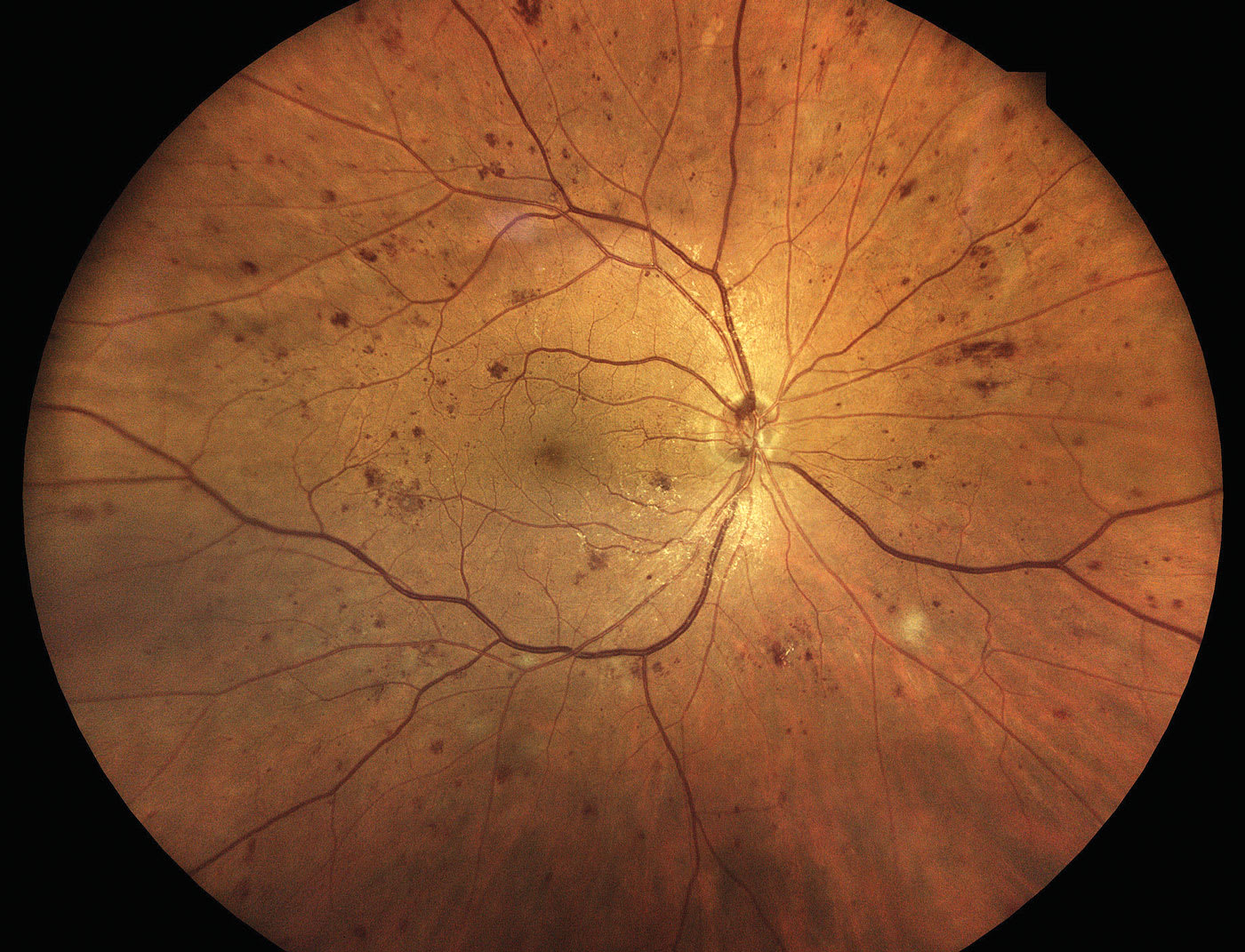
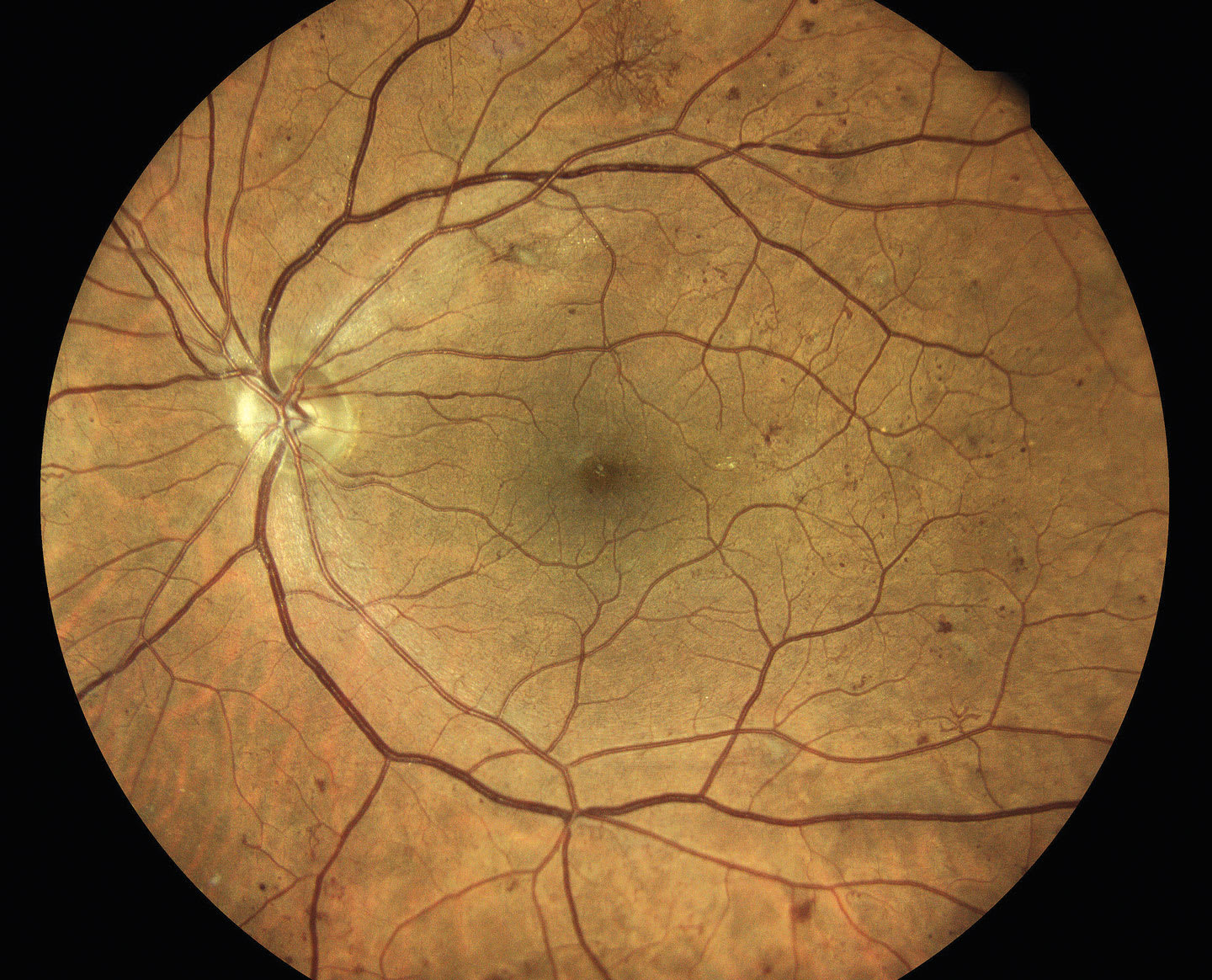
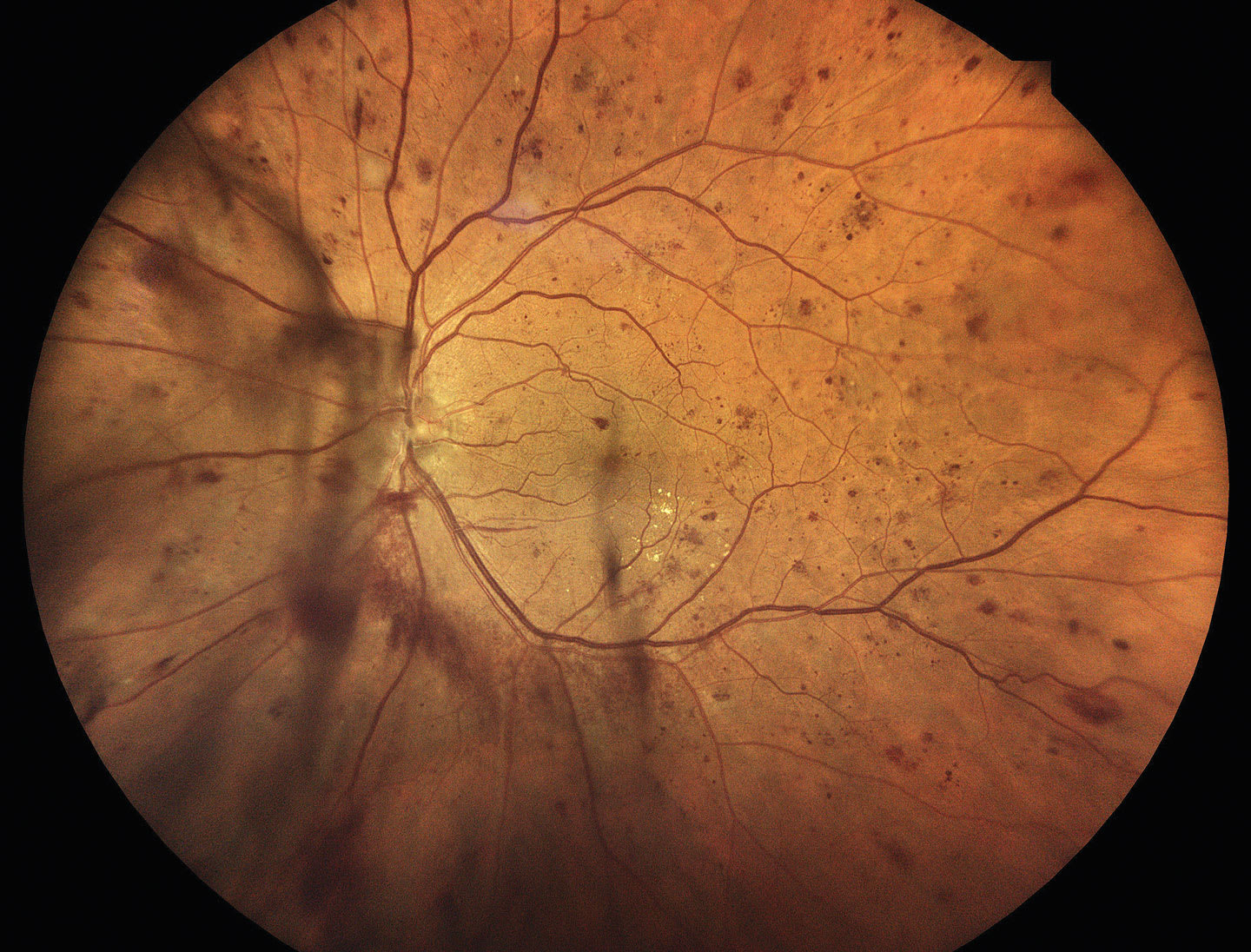
These patients should be referred to a retina specialist for consideration of treatment (ie, panretinal photocoagulation [PRP], as well as serial intravitreal anti-VEGF injections, or a combination of both)8,9 within 1 to 2 weeks. Those who have high-risk PDR should be referred to a retina specialist within 1 to 2 days. Without appropriate treatment, as many as 50% of eyes that have PDR become blind within 5 years.8
Development of Diabetic Macular Edema
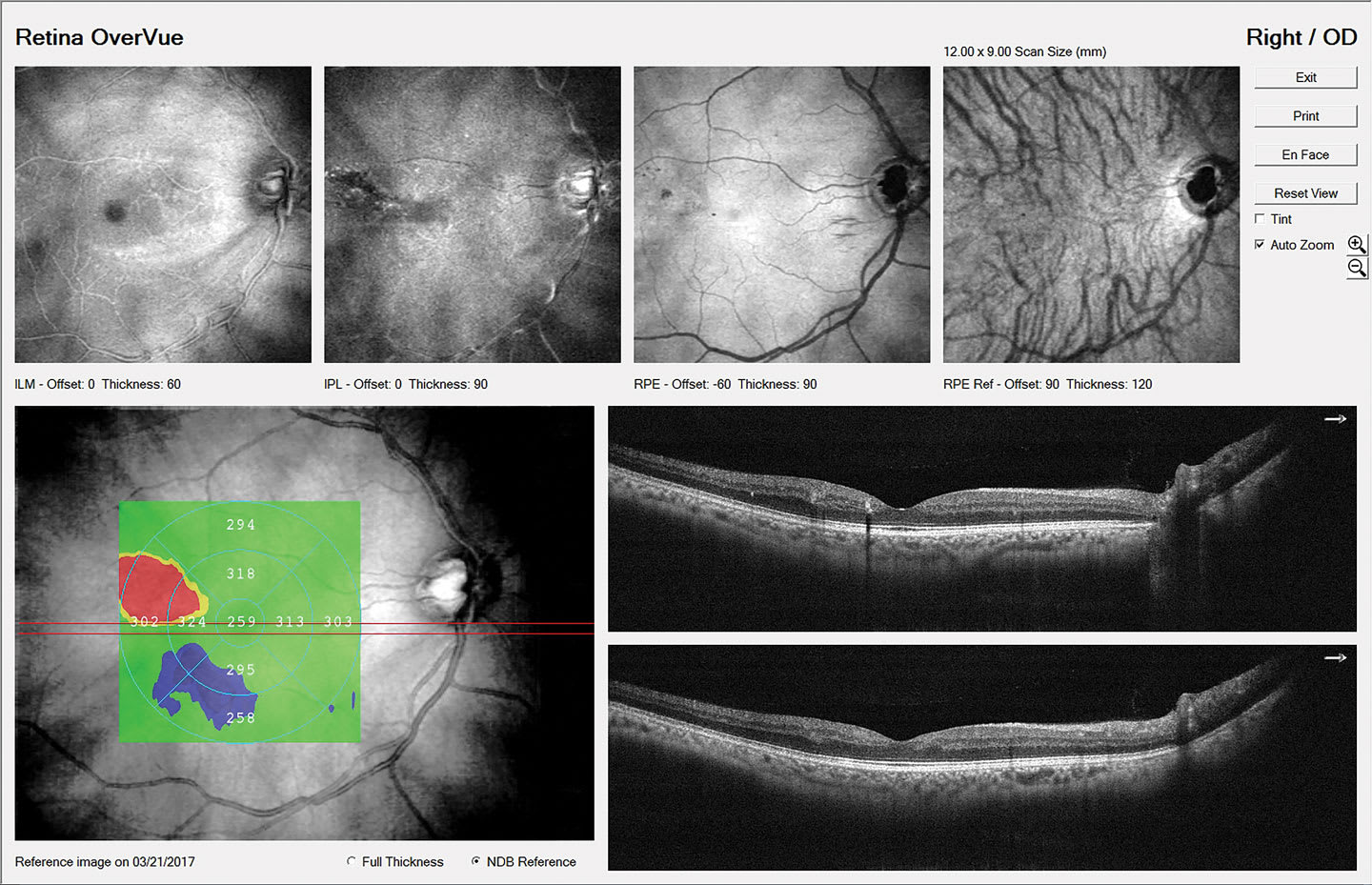
An important caveat regarding staging: Diabetic macular edema (DME) can occur at any stage of retinopathy, from mild NPDR all the way through proliferative disease.10 Diabetic macular edema remains the leading cause of moderate vision loss in patients who have type 2 diabetes, exceeding the vision loss from frank retinopathy. What’s more, it is estimated that nearly 745,000 people, or 3.8% of all patients who have diabetes in the United States, have DME. However, 55% of these patients are unaware they have it, underscoring the importance of detection even in asymptomatic patients.11 Therefore, careful evaluation of the macula for any exudation and/or thickening with optical coherence tomography (OCT) is crucial in all patients who have diabetes.12 Patients who have center-involved DME (ie, the center 1 mm subfield on the OCT showing edema; Figure 7) should be referred to a retinal specialist for consideration of treatment within 2 to 4 weeks.
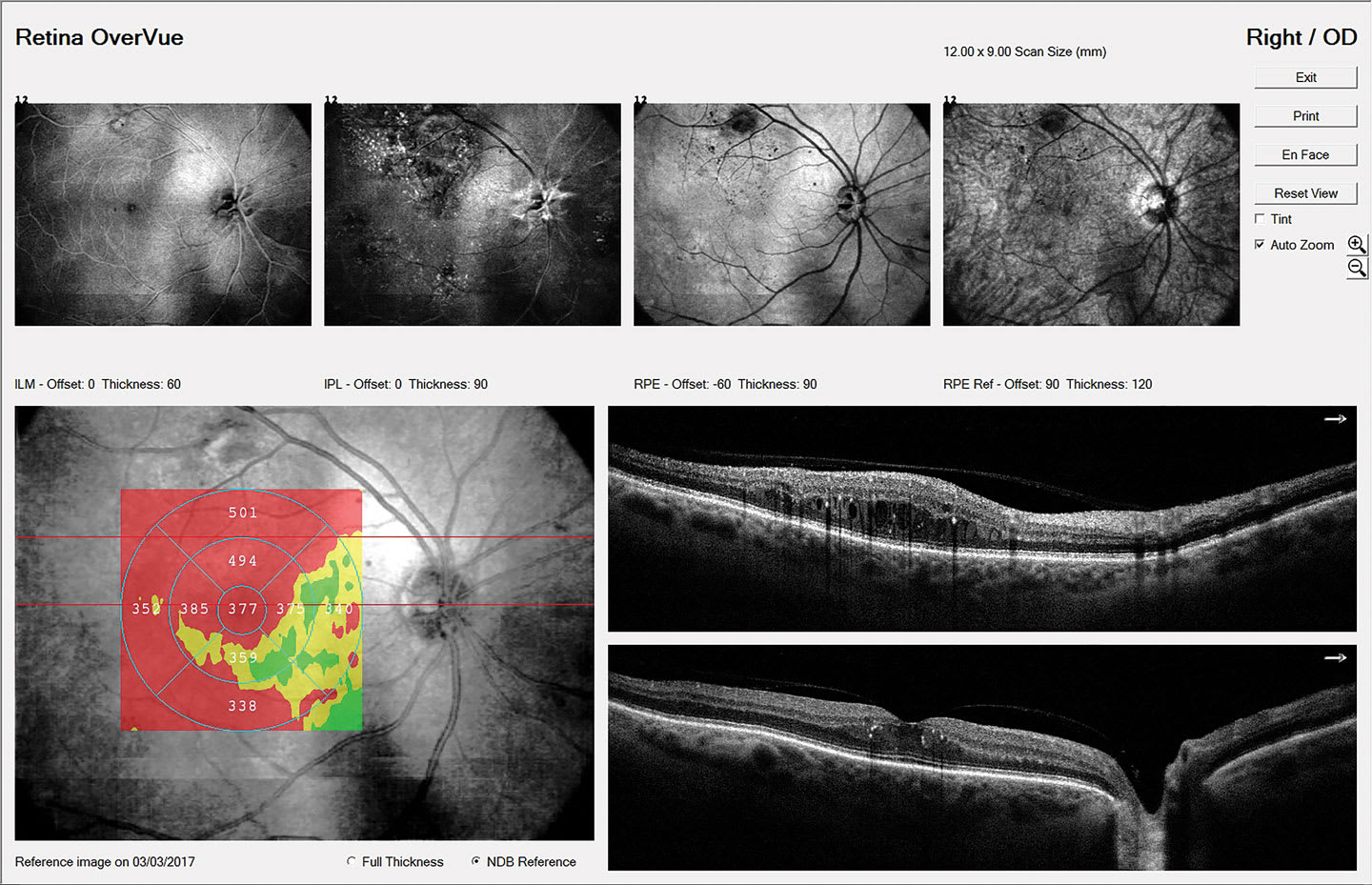
Patients who have non–center-involved DME (ie, the center subfield is spared; Figure 8) should be followed every 3 to 4 months with repeat OCT to monitor for progression.13
New Considerations for Management of Diabetic Retinopathy
There has been much discussion lately regarding the role of glucagon-like peptide-1 receptor agonists (GLP-1 RAs), specifically semaglutide, and their role in diabetic retinopathy. The SUSTAIN-6 study in 2006 was the first to raise the concern regarding increased rates of retinopathy in patients on semaglutide vs other diabetic medications. It showed the rate of diabetic retinopathy in patients treated with semaglutide was 3% vs only 1.8% in patients receiving other treatments. Further, the study authors noted twice the rate of PRP in the semaglutide treated group vs control (2.3% vs 1.2%), as well as slightly more anti-VEGF injections (1% vs 0.8%), vitreous hemorrhages (1% vs 0.4%), and diabetes-related blindness (0.3% vs 0.1%).14
Potential worsening of retinopathy secondary to rapidly improved glycemic control can be a concern for GLP-1 RAs. However, recent studies have suggested such changes are more likely temporary with no long-term detriment to retinopathy progression. Stevens et al showed that semaglutide use was not associated with increased risk of progression of retinopathy, vision loss, and increased number of anti-VEGF treatments over a 3-year period.15,16 Another study showed that patients on GLP-1 RAs had lower rates of DME vs patients taking other diabetic mediations, suggestive of a positive effect of GLP-1 RAs for DME development.17
The AOA’s Comprehensive Adult Eye and Vison Examination, 2nd edition, suggests treating individuals taking GLP-1 RAs as “at risk” patients. These patients should have a baseline dilated eye exam prior to, or shortly after, initiation of treatment, with more frequent examination if they have risk factors, and receive individual risk counseling regarding diabetic retinopathy and their medication. Lastly, collaborative care with other providers managing the patients’ diabetes is recommended.18
A Staging Upgrade?
Researchers are suggesting that the current grading systems are insufficient.19 Current grading systems rely solely on vascular structural changes, and do not account for neurodegenerative retinal changes that often occur prior to the development of clinically evident retinopathy, or systemic health factors that are known to influence the degree of retinopathy.20 Furthermore, newer technologies, such as widefield imaging and OCT angiography, as well as treatments such as anti-VEGF, have been instituted since the development of the grading system, redefining our understanding and approach to diabetic retinopathy. Perhaps with increasing knowledge, the current grading system, as well as this paper, will soon be upgraded to incorporate other important aspects of diabetic retinopathy. OM
References
-
Wilkinson CP, Ferris FL 3rd, Klein RE, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology. 2003;110(9):1677-1682. doi:10.1016/S0161-6420(03)00475-5
-
Yang Z, Tan TE, Shao Y, Wong TY, Li X. Classification of diabetic retinopathy: past, present and future. Front Endocrinol (Lausanne). 2022;13:1079217. doi:10.3389/fendo.2022.1079217
-
He Y, Huang X, Gao S, Zhang M, Zhang J. Does the microaneurysm have to be distinguished from a similar-sized dot hemorrhage? Chin Med J (Engl). 2024;137(20):2504-2506. doi:10.1097/CM9.0000000000003166
-
Klein R, Klein B, Moss S, et al. The Wisconsin epidemiologic study of diabetic retinopathy IX. Four-year incidence and progression of diabetic retinopathy when age at diagnosis is less than 30 years. Arch Ophthalmol. 1989;107:237-43.
-
Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. X. Four-year incidence and progression of diabetic retinopathy when age at diagnosis is 30 years or more. Arch Ophthalmol. 1989;107(2):244-249. doi:10.1001/archopht.1989.01070010250031
-
American Optometric Association. Eye Care of the Patient with Diabetes Mellitus, Second Edition. St. Louis, MO: American Optometric Association; 2019.
-
Flaxel CJ, Adelman RA, Bailey ST, et al. Diabetic Retinopathy Preferred Practice Pattern. Ophthalmology. 2020;127(1):P66-P145. doi:10.1016/j.ophtha.2019.09.025
-
Early photocoagulation for diabetic retinopathy. ETDRS report number 9. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991;98(5 Suppl):766-785.
-
Ferris F. Early photocoagulation in patients with either type I or type II diabetes. Trans Am Ophthalmol Soc. 1996;94:505-537.
-
Perng W, Conway R, Mayer-Davis E, Dabelea D. Youth-onset type 2 diabetes: the epidemiology of an awakening epidemic. Diabetes Care. 2023;46(3):490-499. doi:10.2337/dci22-0046
-
Lee R, Wong TY, Sabanayagam C. Epidemiology of diabetic retinopathy, diabetic macular edema and related vision loss. Eye Vis (Lond). 2015;2:17. Published 2015 Sep 30. doi:10.1186/s40662-015-0026-2
-
Hui VWK, Szeto SKH, Tang F, et al. Optical coherence tomography classification systems for diabetic macular edema and their associations with visual outcome and treatment responses - an updated review. Asia Pac J Ophthalmol (Phila). 2022;11(3):247-257. doi:10.1097/APO.0000000000000468
-
Photocoagulation for diabetic macular edema: Early Treatment Diabetic Retinopathy Study Report no. 4. The Early Treatment Diabetic Retinopathy Study Research Group. Int Ophthalmol Clin. 1987;27(4):265-272. doi:10.1097/00004397-198702740-00006
-
Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/NEJMoa1607141
-
Stevens H, de la Paz M, Cooper B, Bhattacharya R. Long-term use of semaglutide and risk of diabetic retinopathy progression. Endocr Metab Sci. 2024;15:100168. doi:10.1016/j.endmts.2024.100168
-
Cai CX, Nishimura A, Baxter S, et al. Semaglutide and diabetic retinopathy: an OHDSI network study. BMJ Open Diabetes Res Care. 2025;13(6):e005424. Published 2025 Nov 4. doi:10.1136/bmjdrc-2025-005424
-
Muayad J, Loya A, Hussain ZS, et al. Influence of common medications on diabetic macular edema in type 2 diabetes mellitus. Ophthalmol Retina. 2025;9(6):505-514. doi:10.1016/j.oret.2024.12.006
-
American Optometric Association. Comprehensive Adult Eye and Vision Examination, Second Edition. Approved January 20, 2023. https://www.aoa.org/AOA/Documents/Practice%20Management/Clinical%20Guidelines/EBO%20Guidelines/Comprehensive%20Adult%20Eye%20and%20Vision%20Examination%2C%20Second%20Edition.pdf
-
Sun JK, Aiello LP, Abràmoff MD, et al. Updating the staging system for diabetic retinal disease. Ophthalmology. 2021;128(4):490-493. doi:10.1016/j.ophtha.2020.10.008
-
Abramoff MD, Fort PE, Han IC, Jayasundera KT, Sohn EH, Gardner TW. Approach for a clinically useful comprehensive classification of vascular and neural aspects of diabetic retinal disease. Invest Ophthalmol Vis Sci. 2018;59(1):519-527. doi:10.1167/iovs.17-21873