Oculus, Inc. announced that the Pentacam Cornea OCT has been cleared by the US Food and Drug Administration (FDA). According to a company press release, the Pentacam Cornea OCT combines Scheimpflug technology with a pericentric OCT scanning system. In 1 second, cornea specialists are provided with Scheimpflug-based data and ultra high-resolution OCT images of the cornea. Both measurements are taken simultaneously at the same locations, which enables immediate and detailed visualization of anomalies, the company said in the press release.
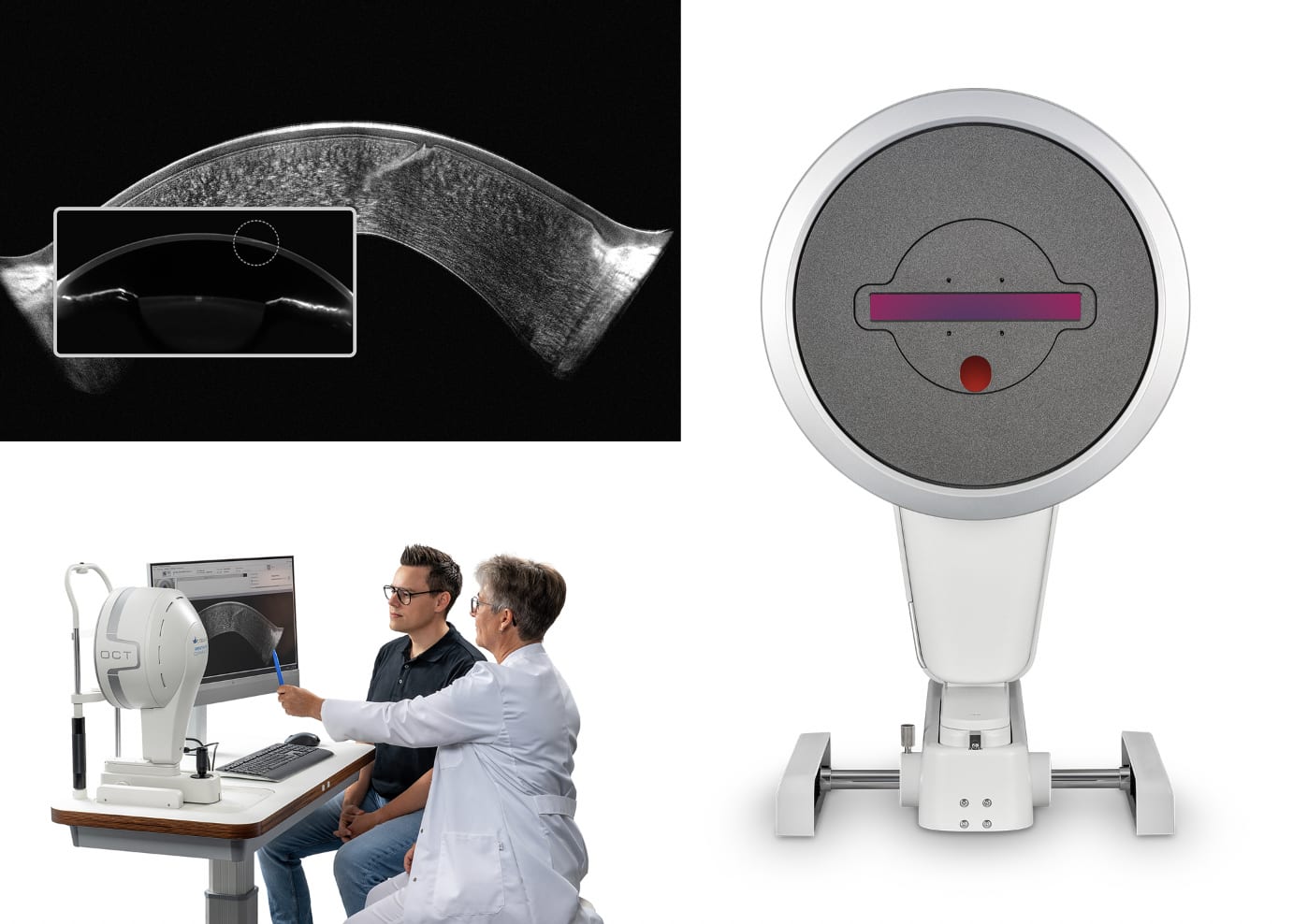
According to Oculus, the combination of Cornea OCT and Scheimpflug imaging allows for comprehensive corneal assessment. The technology is compatible with all existing Pentacam examination data, regardless of the model and software version. This compatibility also simplifies follow-up examinations and progression analysis, the company said.OM